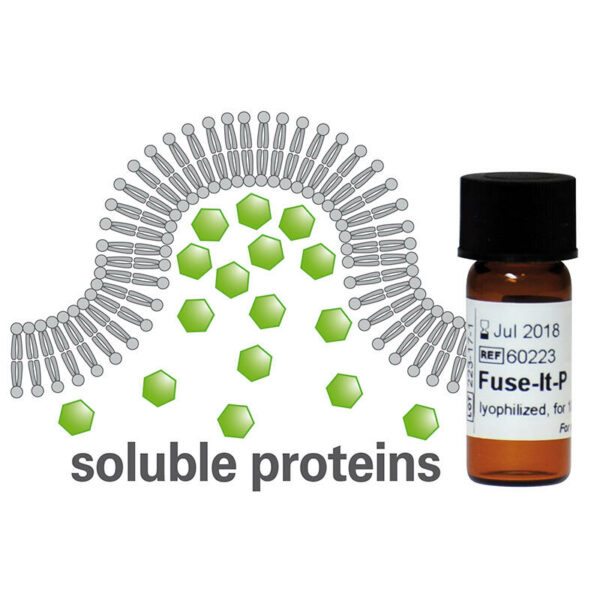
A fusion reagent used to transfer water-soluble proteins into the cytoplasm of living cells
- The lumen of fusogenic liposomes is used as a carrier for water-soluble proteins
- Superior fusion efficiency (up to 80-100%)
- Fusion process completed within 1 - 20 minutes
- Optimized for the transfer of low and intermediate protein amounts to prevent concentration-induced artifacts in cell behavior
- Lyophilized
Applications
- Transfer of labeled proteins or peptides into living cells for functional imaging, speckle analysis, FRAP, or single molecule analysis, etc.
- Incorporation of antibodies or blocking antibodies into living cells
- Blocking, induction, or replacement of protein-induced/regulated signal cascades
- Re-incorporation of proteins into mutant cells
Specifications
| Form | Lyophilized powder |
| Storage | -20 °C |
| ExMax/EmMax | 750/780 nm (infrared) |